

 |
 |
Polysialic Acid (Di/Oligosialic Acid) in the Nervous System |
||||||||||||||||||||||||||
 |
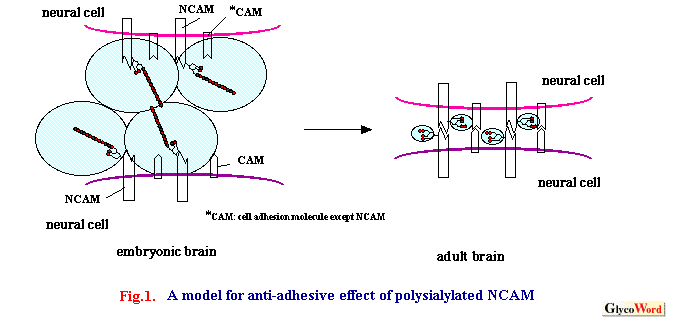
Polysialic acid (PSA) is a polymer of sialic acid whose degree of polymerization (DP) is 8 to 200 Sia residues (1). The most common structure of PSA is the Neu5Ac polymer whose inter-residual linkage is In the nervous system PSA was found in 1982 in the neural cell adhesion molecule (NCAM) derived from chicken and rat embryonic brain. Later, NCAMs derived from the embryonic brain of fish, reptile and amphibian were found to be polysialylated. In 1991, Seki demonstrated that the polysialylated NCAM that had been believed to be restricted in embryonic brain was also present in other restricted regions: the hippocampus and subventricular zone of the forebrain (where neurogenesis continues in adults). In 1992, PSA was also found in developing nervous system of Drosophila. In the same year, the During development from embryo to adult, the protein part of several splicing variants of NCAM is unchanged but the PSA structure dramatically changes from embryonic-type (polysialic acid) to adult type (mono/di(oligo)sialic acid). Since PSA is regulated in a developmental stage-specific manner in brain and nervous system, it was first examined whether PSA regulates the homophilic binding of NCAM. To elucidate the function of PSA, endo-N-acylneuraminidase (Endo-N), which can specifically cleave PSA from the cell surface, and specific antibodies recognizing PSA epitope have been used. Many studies using these approaches have shown that PSA has an anti-adhesive effect in cell-cell and cell-extracellular matrix interactions due to its bulky volume and anionic nature (Fig.1). Furthermore, PSA appears to be a molecule that is involved in the neural cell migration, axonal guidance, fasciculation, synaptogenesis and plasticity via not only homophilic binding of NCAM but also heterophilic binding of other CAMs in a tissue- and stage-specific manner (2). The phenotypes of NCAM knock-out mice are smaller olfactory bulb, reduction of LTP (long-term potentiation) in the CA3 region of the hippocampus, deficits in special learning ability, and abnormality in circadian rhythm compared to wild-type mice. To understand the function of polysialic acid directly, knock-out mice of polysialyltransferases (ST8Sia II (STX) and ST8Sia IV (PST)) responsible for the biosynthesis of PSA on NCAM have been developed. Very recently, mice lacking ST8Sia IV showed decrease in polysialic acid in specific regions of the brain and impairment of LTD (long-term depression) and LTP in the hippocampus CA1 region. However, no decrease in CA3 LTP was observed, unlike for NCAM-deficient mice. Mice lacking ST8Sia II did not impair hippocampus synaptic plasticity but instead resulted in the misguidance of infrapyramidal mossy fibers and the formation of ectopic synapses in the hippocampus. ST8Sia II-deficient mice exhibited higher exploratory drive and reduced behavioral responses. ST8Sia II and ST8Sia IV are expressed in different special and temporal patterns among neural tissues. The ST8Sia II- and ST8Sia IV-deficient mice showed different phenotypes from each other. These results indicate that each enzyme that is able to synthesize the polysialic acid chain plays a different role in the brain. Experiments on double knock-out mice of ST8Sia II and ST8Sia IV are under way. The function of PSA on the voltage-sensitive sodium channel has not been studied so far. |
|||||||||||||||||||||||||
 |
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
Recently, disialic acid and oligosialic acids bearing shorter DP (2 ~ 7) than polysialic acid (DP
|
||||||||||||||||||||||||||
| Chihiro Sato (Nagoya University) | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Nov. 30, 2004 | ||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||