 |
Universality of mannose trimming
All the eukaryotes ever analyzed and some prokaryotes have N-glycans. Since the N-glycans of prokaryotes are quite different from those of eukaryotes, the origin and function may differ from each other. Biosynthesis of N-glycans in eukaryotes starts when a high mannose-type tetradecasaccharide transfers en bloc to protein. The mannoses are trimmed one by one from the glycan, and the glycan become a heptasaccharide with five mannoses in metazoan. Several pathways are possible for the mannose trimming. However, it seems that the main trimming pathway is almost single in metazoans from planaria to humans. There may be some control factor for change in the pathway. Specifically, it suggested that the selected isomers of high mannose-type are required for some physiological phenomenon. It is probable that those isomers participate in the intracellular transport of glycoproteins. Moreover, it is suggested that the specific isomer of eight mannoses is important in the quality control of glycoproteins.
Diversity of complex-type glycans
It seems that the complex-type glycans appeared in the metazoans and were not seen in the protozoans. Since there is scarce data on the glycan structures in lower metazoans, for example, sponges (poriferan) or jellyfish and hydra (cnidarians), the exact origin of the complex-type is not known yet. While the high mannose-type glycans are universal, the complex-type glycans are extremely diverse. Although the trimannose-core structure (pentasaccharide with three mannoses) is common, other portions differ considerably. This fact leads us to expect that there is some relation between the diversity of glycan structures and the biodiversity of multicellular organisms. The complex-type glycans such as biantenna and triantenna with LacNAc are not found except in vertebrates, although they are quite common in vertebrates. Invertebrates seem not to have the type-2-lactosamine-structure (Gal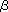 1-4GlcNAc). However, to know the exact origin of the type-2-lactosamine-structure, research on the glycan of sea squirt (tunicate) or amphioxus (cephalochordate) is required. They are the organisms just before vertebrates. On the other hand, the type-1-lactosamine structure (Gal 1-4GlcNAc). However, to know the exact origin of the type-2-lactosamine-structure, research on the glycan of sea squirt (tunicate) or amphioxus (cephalochordate) is required. They are the organisms just before vertebrates. On the other hand, the type-1-lactosamine structure (Gal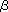 1-3GlcNAc) is found in cephalopods, such as squid and octopus, and the LacdiNAc structure (GalNAc 1-3GlcNAc) is found in cephalopods, such as squid and octopus, and the LacdiNAc structure (GalNAc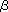 1-4GlcNAc) exists widely from insects to vertebrates. Although it seems that the nematodes have peculiar complex-type glycans, complete structures have not been determined yet. Complex-type glycans make groups of structures peculiar to each clade according to variations of such extension structures, branch structures, and the acid residues of non-reducing ends including the sialic acids. 1-4GlcNAc) exists widely from insects to vertebrates. Although it seems that the nematodes have peculiar complex-type glycans, complete structures have not been determined yet. Complex-type glycans make groups of structures peculiar to each clade according to variations of such extension structures, branch structures, and the acid residues of non-reducing ends including the sialic acids.
Pauci-mannose-type as anti-diversity
Although the pauci-mannose-type glycans are not commonly observed in vertebrates, it is a very ordinary type in invertebrates. The biosynthetic pathway of the pauci-mannose-type glycans may be stationed to interfere with the process to the complex-type glycans. That is, the pathway can be regarded as a route which leads to monotonous structures and hinders diverse formation of glycans. A key to the processing to the pauci-mannose pathway is an enzyme called processing N-acetylglucosaminidase, which is a membrane protein. This enzyme hydrolyzes the sugar moiety transferred by N-acetylglucosaminyltransferase-I. If the complex-type is involved in morphogenesis and the pauci-mannose-type interrupts the processing to the complex-type, the complex-type glycans ought to be discovered at the time of morphogenesis and interchanged to pauci-mannose-type in adult organisms. The report on the expression of processing N-acetylglucosaminidase in the developmental stages of the fruit fly brain supports this expectation.
|
|
|
| References |
(1) |
Natsuka S: Comparative biochemical view of N-glycans. Trends Glycosci. Glycotechnol., 17, 229-236, 2005 |
|
(2) |
Angata T, Varki A: Chemical diversity in the sialic acids and related alpha-keto acids: an evolutionary perspective. Chem. Rev., 102, 439-469, 2002
|
|
|



