Eiji Miyoshi
Osaka University, Graduate School of Medicine, Department of Molecular Biochemistry & Clinical Investigation
It is a well-known fact that oligosaccharide structures of glycoproteins and expression of glycosyltransferases change in malignant transformation. In approximately 300 kinds of these enzymes, N-acetylglucosaminyltransferase V (GnT-V) is one of the most important glycosyltransferases which are involved in tumor metastasis. Recent studies of GnT-V-deficient mice have demonstrated that GnT-V is essential for tumor metastasis as well as tumor growth 1. Mammary tumor growth and metastases induced by the polyomavirus middle T oncogene were considerably less in GnT-V- deficient mice than in transgenic littermates expressing GnT-V. On the other hand, since expression of GnT-V was elevated at the stage of chronic hepatitis as well as in the metastatic lesion of a rodent model of hepatocarcinogenesis 2, GnT-V is assumed to be involved in the early event of carcinogenesis. The most important biological phenomenon in the early phase of carcinogenesis is expected to be immortalization and tumor angiogenesis. In 1993, Pierce’s and Taniguchi’s groups performed protein purification and cDNA cloning of GnT-V. After 10 years, a unique function of GnT-V as an angiogenesis inducer was found 3. The mechanisms for GnT-V-induced tumor angiogenesis were due to completely different functions from those of glycosyltransferases. In general, glycosyltransferases are located in the Golgi apparatus and involved in the oligosaccharide modification of glycoproteins/glycolipids. However, a glycosyltransferase also exists in the serum of patients and it is reported to be a biomarker for certain diseases. Certain proteases digest a glycosyltransferase and secrete into the condition medium of cells. This type of glycosyltransferase is the soluble form. In the case of GnT-V, the soluble form of the enzyme has the biological function of angiogenesis. The basic domain of GnT-V, KRKRKK, which is located in the 264-269 amino acid sequence, promotes a release of bFGF from the extra cellular matrix, resulting in tumor angiogenesis (see Fig. 1). Many kinds of cytokines/growth factors attach to proteoglycans such as heparansulfates in the extra cellular matrix. If the basic domain of GnT-V is associated with the release or activation of these factors, the soluble form of GnT-V could have a variety of biological functions. Furthermore, the identification of a protease which is responsible for cleavage and secretion of GnT-V may provide new insights in glycobiology.
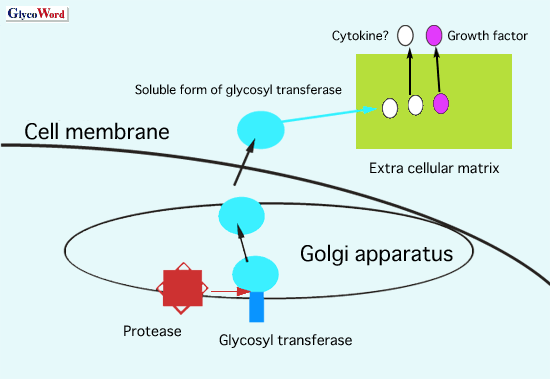
Fig. 1 logical functions of a soluble from of glycosyl transferase.