
Izuru Kawamura
Professor, Yokohama National University
He received his Ph.D. in Engineering from Yokohama National University in 2007. After serving as a research faculty member at Yokohama National University and as a visiting researcher at the University of Guelph, Canada, he was appointed Assistant Professor at Yokohama National University in 2012, promoted to Associate Professor there in 2013, and has been a Professor at the Faculty of Engineering, Yokohama National University, since 2024. His research interests include biophysical chemistry and solid-state nuclear magnetic resonance spectroscopy of biological systems. He received the Progress Award from the Nuclear Magnetic Resonance Society of Japan in 2023.

Noriko Kanai
Noriko received her Ph.D. in Chemistry from Yokohama National University in 2023. Following her work as a Japan Society for the Promotion of Science (JSPS) Postdoctoral Fellow, she became and is currently an Assistant Professor at the Faculty of Environment and Information Sciences, Yokohama National University. Since June 2024, she has also been conducting research as a JSPS Overseas Research Fellow at the School of Science and Hawkesbury Institute for the Environment, Western Sydney University, where she has been a Visiting Fellow since 2022. Her current research focuses on nanocellulose derived from agricultural/food waste and on the structural analysis of cell wall extracts from various biomass sources.
Food waste and food loss account for nearly 10% of global greenhouse gas emissions, seriously contributing to accelerating climate change1. Coffee is one of these foods that create waste. Coffee is an agricultural product with high global consumption and large distribution volumes. Large quantities of spent coffee grounds (SCGs) are generated during coffee processing and consumption; SCGs are thus a common form of food waste. The majority of SCGs is disposed of in landfills2, and approximately 11.14 million tons of SCG waste were generated between 2022 and 2023 according to statistics from the International Coffee Organization3. SCGs are a promising biomass resource because they contain various components that can be used as raw material for fuels, biopolymers, and other products4. Furthermore, SCGs are a model waste product for the “cross-economy,” a concept that redefines the use of resources and redesigns resource circulation among different industries and technological fields5. In this paper, we outline the manufacturing process, structure, and physical properties of a novel nanocellulose derived from SCGs.
Cellulose is a renewable immobilized product of carbon dioxide obtained from the atmosphere. Cellulose nanofibers (CNFs) are manufactured from various biomass resources, such as wood, by mechanical and chemical defibration of cellulose into nanometer-scale fibrous structures6. Holocellulose nanofibers (HCNFs) are CNFs that are manufactured from lignin-removed holocellulose, a raw material that retains the cellulose and hemicellulose fractions7. Furthermore, HCNFs have been applied in food packaging materials8, multicomponent composite films9, and aerogels, which are lightweight porous materials10. The physical properties and structure of the resulting nanocellulose widely vary depending on the type of raw biomass and its polysaccharide composition.
SCGs are waste resources that contain cellulose (~10%), hemicellulose (~40%), lignin (~30%), and lipids (~15%), regardless of the roasting level or type of coffee beans11. The content of these SCG constituents was confirmed using 1H/13C solid-state nuclear magnetic resonance (NMR)12. Additionally, the main monosaccharides in SCGs are mannose (~29%), galactose (~11%), glucose (~11%), and arabinose (~2.1%). The main hemicellulosic components in SCGs are galactomannan and arabinogalactan11. The basic galactomannan structure includes a (1→4)-β-D-mannopyranose backbone with (1→6)-α-linked α-D-galactopyranose side chains13. The galactomannan derived from coffee beans has a low substitution rate of (1→6)-α-linked α-D-galactopyranose for (1→4)-β-D-mannopyranose backbone (approximately 1–2%) and has a nearly linear mannan structure14,15. Mannan exists in two crystalline polymorphs: types I and II. Mannan I, which is characterized by a crystal structure analogous to cellulose I, exhibits higher thermal stability; therefore, mannan I is considered to be predominant in SCG.

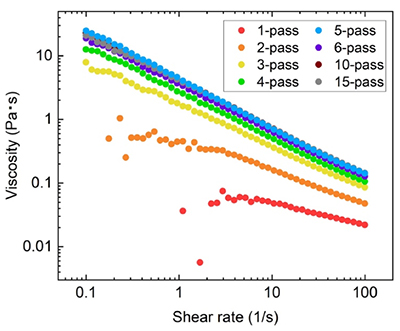
In this study, we established a process for manufacturing HCNFs using SCGs as the raw material. We used dried SCGs collected from offices and cafes within Kamakura City in Japan as the starting raw material. First, the lipids in the SCGs were removed using an acetone/water (9:1 v/v) solvent, followed by multistage delignification with a sodium chlorite/acetic acid system at 75℃ (Wise method). This pretreatment yielded a holocellulose powder from the raw SCGs at approximately 50%, and mannan and cellulose were confirmed as the main components of the powder. The obtained holocellulose powder was dispersed in water to a concentration of 2%. Mechanical defibration using a high-pressure wet jet mill was performed multiple times to convert the holocellulose powder into SCG-derived HCNFs (Fig. 1)16. We determined the rheological properties of the HCNF dispersed in water while varying the number of passes the material made through the jet mill; approximately five passes of the SCG-derived holocellulose through the jet mill yielded a high-viscosity dispersion. Additionally, this dispersion exhibited thixotropic behavior: the viscosity decreased with increasing shearing rate (Fig. 2). Based on this result, it was determined that the five-pass treatment was optimal for manufacturing HCNFs in this system. We previously used 2,2,6,6-tetramethylpiperidine-1-oxyl radical (TEMPO)-mediated oxidation treatment17 to prepare TEMPO-oxidized CNFs from SCGs18. However, the low yield of the obtained CNFs, at approximately 10%, due to the inherently low cellulose content of SCGs, posed a major challenge to CNF production. In contrast, by taking into account the properties of the SCGs, including those of the hemicellulose fraction, the method of manufacturing SCG-derived HCNFs used in this study effectively increased yield.
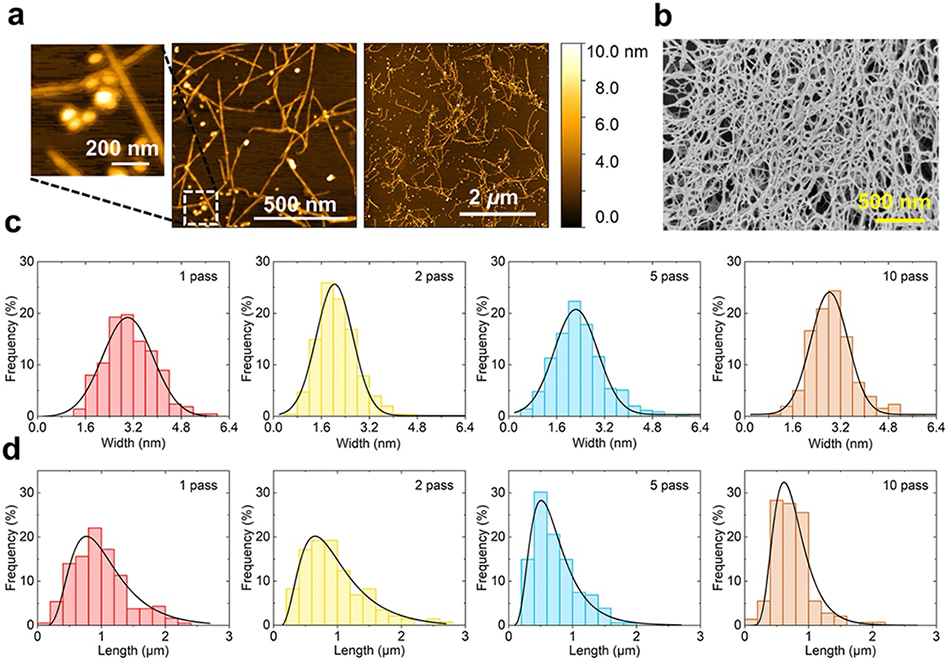
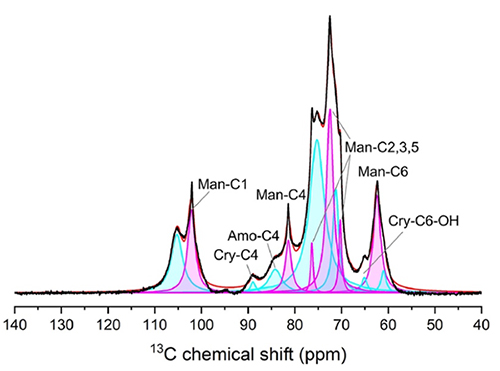
We observed the morphology and structure of the obtained HCNFs using atomic force microscopy (AFM) and scanning electron microscopy as shown in Fig. 3a and Fig. 3b. After the first pass of treatment, defibration progressed to approximately 3 nm, which corresponded to the theoretical microfibril width. Furthermore, the fiber width remained nearly constant after 2, 5, and 10 passes: the fiber width did not change as the number of passes increased16. The mean HCNF length was approximately 1 µm after 1 or 2 passes and decreased to approximately 0.7 µm after 5 or 10 passes (Fig. 3c and 3d). AFM observation revealed mannan-derived crystalline particles with a diameter of approximately 5–10 nm, in addition to the fibrous structure. The 13C cross-polarization magic-angle spinning NMR spectrum of the freeze-dried HCNF product simultaneously showed a cellulose-derived signal and a slightly stronger mannan-derived signal (Fig. 4). The line width of the mannan-derived signal was narrower than that of the cellulose-derived signal. This narrow mannan-derived signal was attributed to type I mannan, which is highly crystallizable. Moreover, the peaks corresponding to the major crystal planes of type I mannan were detected in the powder X-ray diffraction pattern of holocellulose, at approximately 2θ = 16°, 20°, and 25°. These findings demonstrated that the SCGs-derived HCNFs were mainly composed of CNFs with a width of approximately 3 nm as well as numerous mannan particles with high crystallinity.
We found that when HCNFs that were dispersed in water up to a concentration of approximately 5% were freeze-dried and then redispersed in water using ultrasonication combined with other treatments, the dispersion state of the HCNFs was restored. Typical CNFs do not display this behavior, as CNFs tend to form strong aggregates upon drying. This finding thus indicates that the storage, transportation, and reuse properties of HCNFs are superior to those of typical CNFs. We are now currently applying insights from surface modification treatments, such as fluorescent modification19 and hydrophobization20,21 of CNFs derived from hop stems, as well as analyzing the hemicellulose structure and other properties of SCG-derived HCNFs.
We developed a process for manufacturing mannan-rich, microfibril-width HCNFs derived from SCGs, food waste, through mechanical defibration. The rheological properties of the obtained HCNFs were determined, indicating the high crystallinity of mannan. We also increased the HCNFs yield compared with that achieved in our prior study. Additionally, the redispersibility of the SCGs-derived HCNFs was superior to that of typical CNFs, indicating the developed HCNFs are a novel nanocellulose material with unique properties.
Plant-based agricultural and food waste such as fruit peels and leaves have attracted attention as promising raw materials for nanocellulose production. The use of food and agricultural wastes as an emulsion stabilizer for Pickering emulsions has been widely studied22. SCGs may serve as an important biomass resource that contributes to the development of a circular economy in society, as SCGs are generated in large quantities and require minimal processing.