Tadashi Suzuki
Chief Scientist, Glycometabolic Biochemistry Laboratory; RIKEN Pioneering Research Institute (PRI)
Dr. Suzuki obtained a Ph. D. (Doctor of Science) degree in 1997 from the Department of Biochemistry and Biophysics, Graduate School of Science, University of Tokyo (Advisors: Prof. Yasuo Inoue/Assoc. Prof. Yasufumi Emori). From July 1997 to 2001, he was a Post-doctoral Fellow/Research Scientist/Research Assistant Professor at State University of New York at Stony Brook (Prof. William J. Lennarz). In December 2001 he came back to Japan as a Researcher of the Precursory Research for Embryonic Science and Technology (PRESTO), Japan Science and Technology Corporation (JST). From February 2002 to January 2004, he also served as an RCF Assistant Professor at the Graduate School of Science, University of Tokyo, and in January 2004, he became Visiting Associate Professor at Osaka University Graduate School of Medicine. In October 2007, he became a Team Leader of the Glycometabolome Team, Systems Glycobiology Research Group, RIKEN, and has held his current position since April 2018.
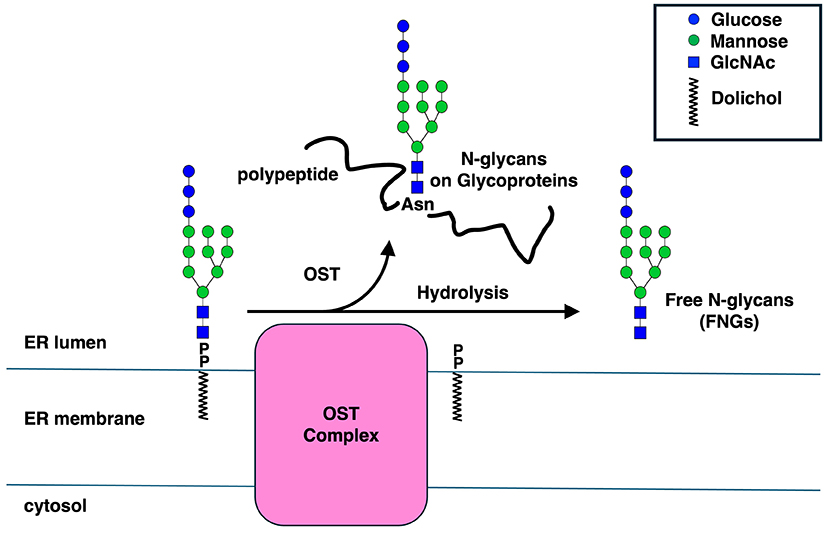
Asparagine (N)-linked glycans on proteins are one of the most common co- and post-translational modifications found not only in eukaryotes but also in bacteria and archaea. This reaction is carried out by enzymes called “oligosaccharyltransferases” (OSTs). While the active subunit often functions alone in bacteria and archaea, different subunit structures are known to exist in eukaryotic cells depending on the species. Recently, it has become clear that OSTs not only transfer glycans to proteins but also generate free glycans by hydrolysis of donor substrates, i.e., dolichol-linked oligosaccharides (DLOs). This hydrolysis reaction is significantly enhanced in mammals compared to budding yeast, suggesting that OSTs have evolved to more efficiently hydrolyze DLOs to form free N-glycans. Here, we provide an overview of what is known about OSTs and propose a hypothesis regarding the significance of their hydrolysis reaction.
Oligosaccharyltransferases (OSTs) transfer pre-assembled glycans (14 sugars in budding yeast [Saccharomyces cerevisiae] and humans [Glc3Man9GlcNAc2; see https://www.glycoforum.gr.jp/article/06A5.html]) onto a defined sequence (called a sequon: Asn-Xaa-Ser/Thr, where Xaa is any amino acid except Pro) on lipids. In eukaryotes, OSTs are localized to the endoplasmic reticulum (ER) and transfer oligosaccharides to proteins, both co-translationally and post-translationally, during translation. Glycosylation has a profound effect on the physical properties as well as physiological functions of cognate proteins, and genetic mutations in these processes are known to cause genetic disorders collectively known as congenital disorders of glycosylation1. Recent advances in cryo-EM techniques have clarified insights into the substrate recognition and reaction mechanisms of OSTs (please refer to recent reviews2,3 for details).
An OST is a complex composed of many subunits in mammals and budding yeast, but some species appear to possess only the catalytic subunit. For example, Leishmania major has a functioning OST that lacks subunits other than the catalytic subunit. On the other hand, the OST of this organism is known to possess multiple catalytic subunits (LmSTT3A-D), suggesting that maintaining OSTs with different properties is likely important for Leishmania major4.
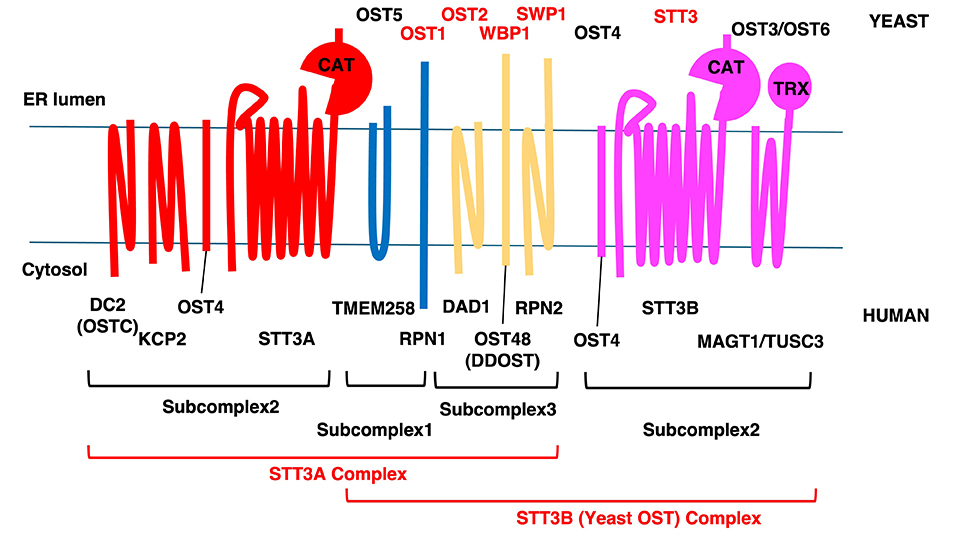
OST subunits have been extensively characterized in mammals and budding yeast. Budding yeast is known to have eight subunits organized into three subcomplexes (Fig. 1). Subcomplex 1 is composed of OST1 and OST5; subcomplex 2 is STT3, OST4, and OST3/6; and subcomplex 3 is WBP1, SWP1, and OST2. In budding yeast, STT3, WBP1, SWP1, OST1, and OST2 are essential genes, while OST3, OST4, OST5, and OST6 are not. OST3 and OST6 are interchangeable functional paralogs, and it is speculated that the CXXC motif on OST3/6 increases the probability of STT3 encountering the sequon by forming a disulfide bond with the substrate protein5. While STT3 is shown to be the catalytic subunit responsible for the oligosaccharide-transfer activity, the precise functional importance of the other subunits remains relatively unclear.
In recent years, OSTs have been shown to possess hydrolytic activity for DLOs, leading to the release of free N-glycans (FNGs; free oligosaccharides structurally related to N-glycans) (Fig. 2)6,7. It has been shown in budding yeast that the hydrolytic activity of OSTs is clearly discernable from its oligosaccharyl transfer activity8. In mammals, OST hydrolytic activity appears to be carried out exclusively by the STT3B complex9. Furthermore, it has recently been shown that the hydrolytic activity of budding yeast OST is enhanced under ER stress conditions where increased level of misfolded proteins in the ER is observed10. We propose that FNGs may function as chaperones to prevent protein aggregation or promote protein folding or both10. The positive effects of N-glycans on preventing protein aggregation or promoting protein folding have been biochemically verified in previous studies11,12.
Interestingly, the hydrolytic activity of mammalian OST is significantly higher than that of budding yeast OST (the amount of FNGs produced per cell by mammalian OST is approximately 10,000-fold higher than that of budding yeast OST7). This suggests that mammalian OSTs evolved this higher hydrolytic activity for a reason. In this regard, the presence of serum FNGs is noteworthy13. It has been shown that OST is, at least in part, responsible for the secretion of these FNGs14. FNGs are also found in salmon serum, suggesting that serum-free glycans are conserved among vertebrates15.