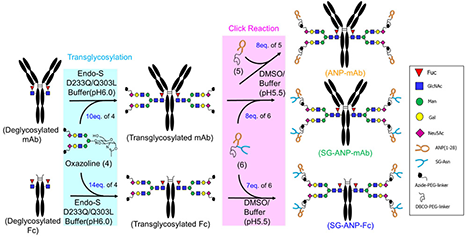
Glyco-engineering is an advanced technology that artificially designs and modifies the composition and structure of glycans bound to biomolecules such as peptides and proteins. In the biopharmaceutical field, a wide range of approaches have been investigated that aim to enhance functionality, enhance target specificity, and reduce side effects by modulating drug properties such as stability, efficacy, safety, and pharmacokinetics. Particularly, chemoenzymatic glycoengineering of monoclonal antibodies using endo-beta-N-acetylglycosaminidase (ENGase) is known to provide useful tools such as linkers for site-specific conjugation of drugs to antibodies. This review provides recent updates of drug discovery research using chemoenzymatic approaches and developments to overcome challenges in large-scale production....and more
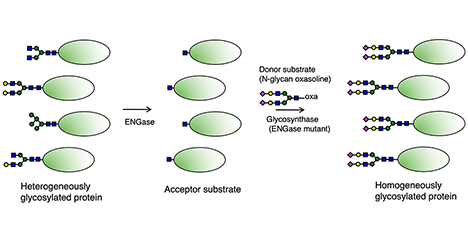
Glycans play important roles in protein function, stability, intermolecular interaction, and so on. These are highly related to glycan structures in many cases. However, the structure of the glycan attached to the specific glycosylation site usually varies in each molecule of glycoprotein, and this variation gives rise to the heterogeneity and functional differences between glycoproteins. Therefore, glycan remodeling aims to increase the homogeneity and strengthen the function and stability of the glycoprotein by attaching glycans of specific structure to the glycosylation sites of the glycoprotein. The glycan remodeling techniques have been widely developed, and one of them is the endo-β-N-acetylglucosaminidase (ENGase, EC 3.2.1.96)-based method. In connection with this, this review outlines the basic knowledge of glycan remodeling and the recently identified novel ENGases that recognize the non-reducing end of N-glycans and can hydrolyze multibranched complex type N-glycans....and more
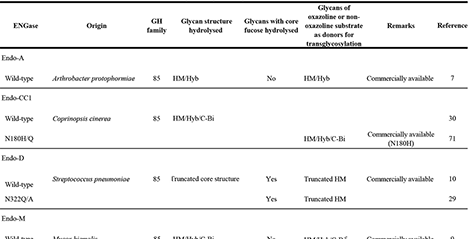
Endo-β-N-acetylglucosaminidase (ENGase) is an enzyme widely distributed among various organisms and known to hydrolyze N-glycans. Recent studies have demonstrated that strategic modification of its catalytic residues enables its conversion into a glycosynthase, thereby facilitating the chemoenzymatic remodeling of N-glycan structures.
In this series, we will discuss recent progress in glycoengineering-related ENGase research by leading experts in each field....and more